A solution containing a mixture of 0.05 M NaCI and 0.05 M Nal is taken. ( Ksp of AgCl = 10^-10 and Ksp of AgI = 4 × 10^-16 ). When AgNO3 is added to such a solution

Why common salt is added to precipitate out soap form the solution during its manufacturing ? - YouTube
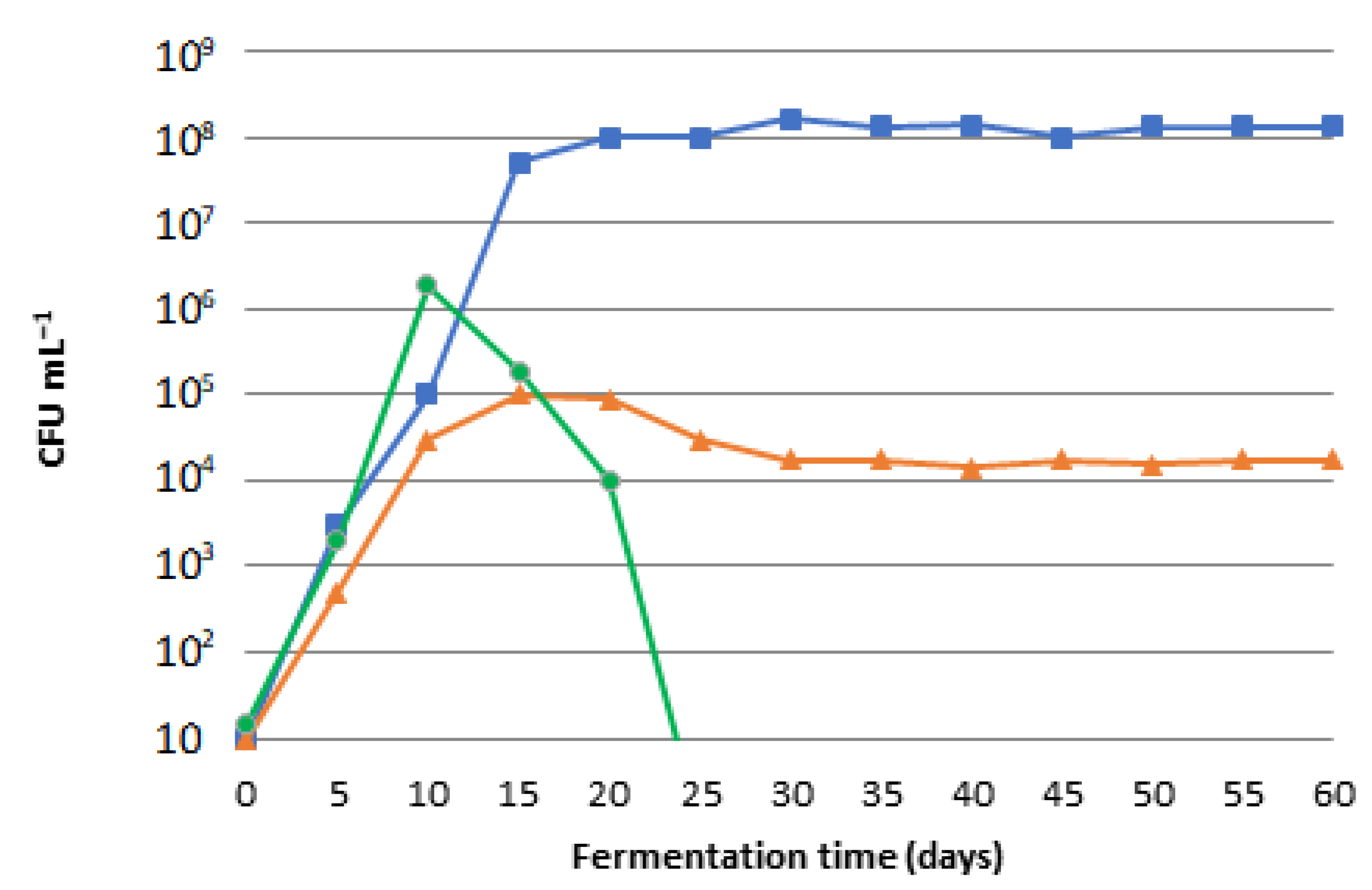
Fermentation | Free Full-Text | Production and Maturation of Soaps with Non-Edible Fermented Olive Oil and Comparison with Classic Olive Oil Soaps

SOLVED: Lab 18: Intermolecular Forces and the Synthesis of Soap Date: Lil Name: Purpose: To examine the effects of intermolecular forces on the solubility of soap in different types of water. Contents