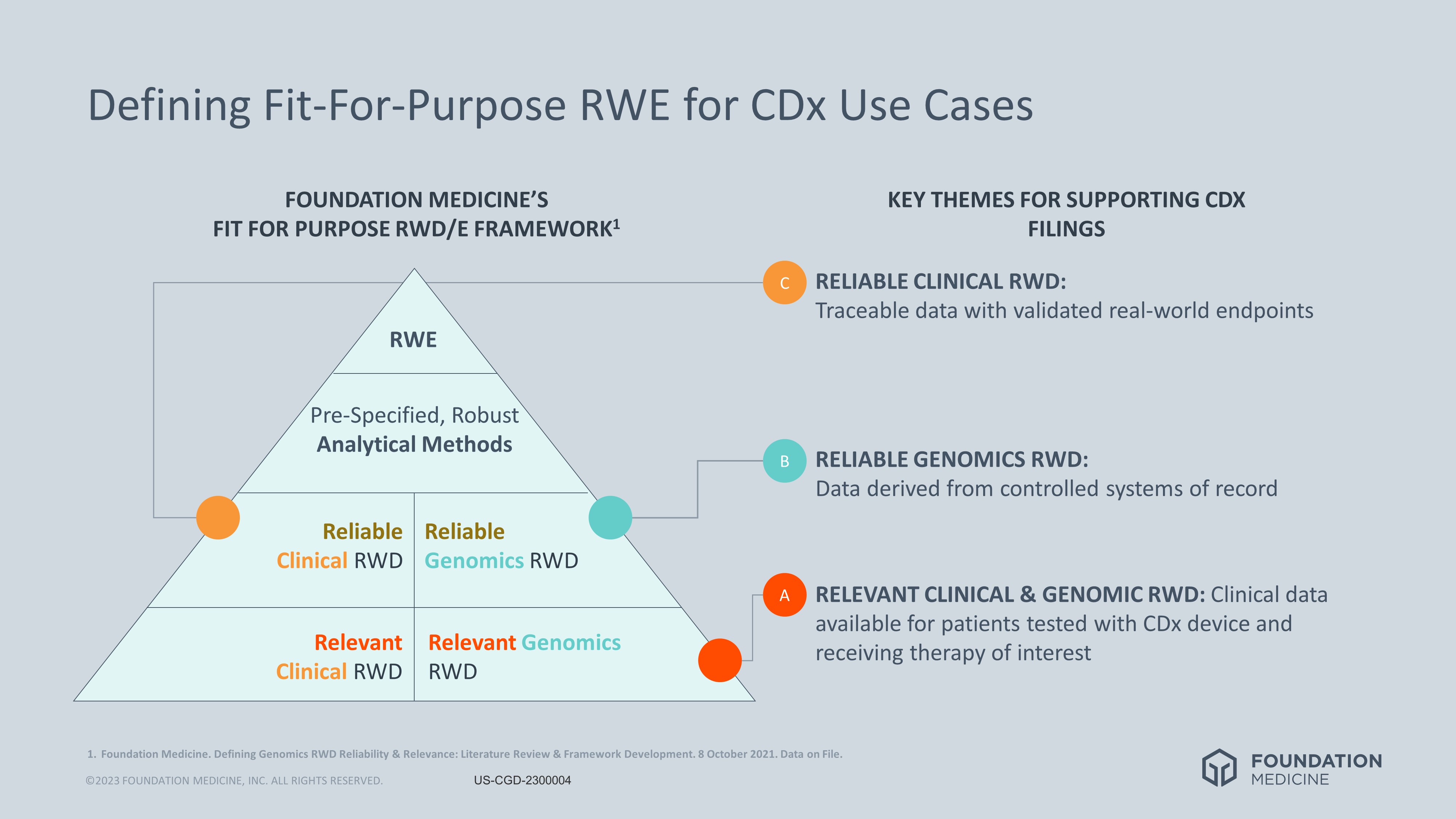
Foundation Medicine Unveils Strategy to Support Cancer CDx Filings with RWE from Clinico-Genomic Database | Precision Medicine Online

Matthew Nelson on LinkedIn: Great achievement by the Foundation Medicine! Check out the latest FDA CDx…
![Workflow for TMB assessment using the FoundationOne CDx assay [63].... | Download Scientific Diagram Workflow for TMB assessment using the FoundationOne CDx assay [63].... | Download Scientific Diagram](https://www.researchgate.net/publication/334081089/figure/fig2/AS:774610417090560@1561692923137/Workflow-for-TMB-assessment-using-the-FoundationOne-CDx-assay-63-ExAC-Exome.png)
Workflow for TMB assessment using the FoundationOne CDx assay [63].... | Download Scientific Diagram
FoundationOne®CDx Technical Information Foundation Medicine, Inc. 150 Second Street, Cambridge, MA 02141 Phone: 617.418.2200 I

UnitedHealthcare to offer coverage for Foundation Medicine CDx tests for multiple cancer types | LabPulse.com

Foundation Medicine and Its Collaborators Announce Acceptance of 21 Abstracts at the 2023 American Society of Clinical Oncology (ASCO) Annual Meeting









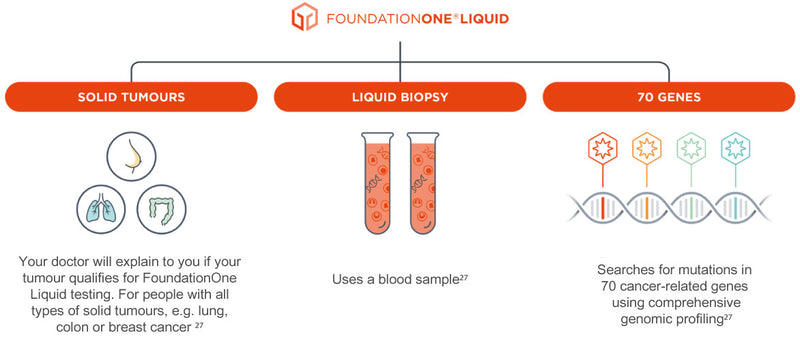
![Foundation Medicine review - 7 facts you should know [DECEMBER 2021] Foundation Medicine review - 7 facts you should know [DECEMBER 2021]](https://nebula.org/blog/wp-content/uploads/2021/12/Foundation-Medicine-report-1024x494.png)